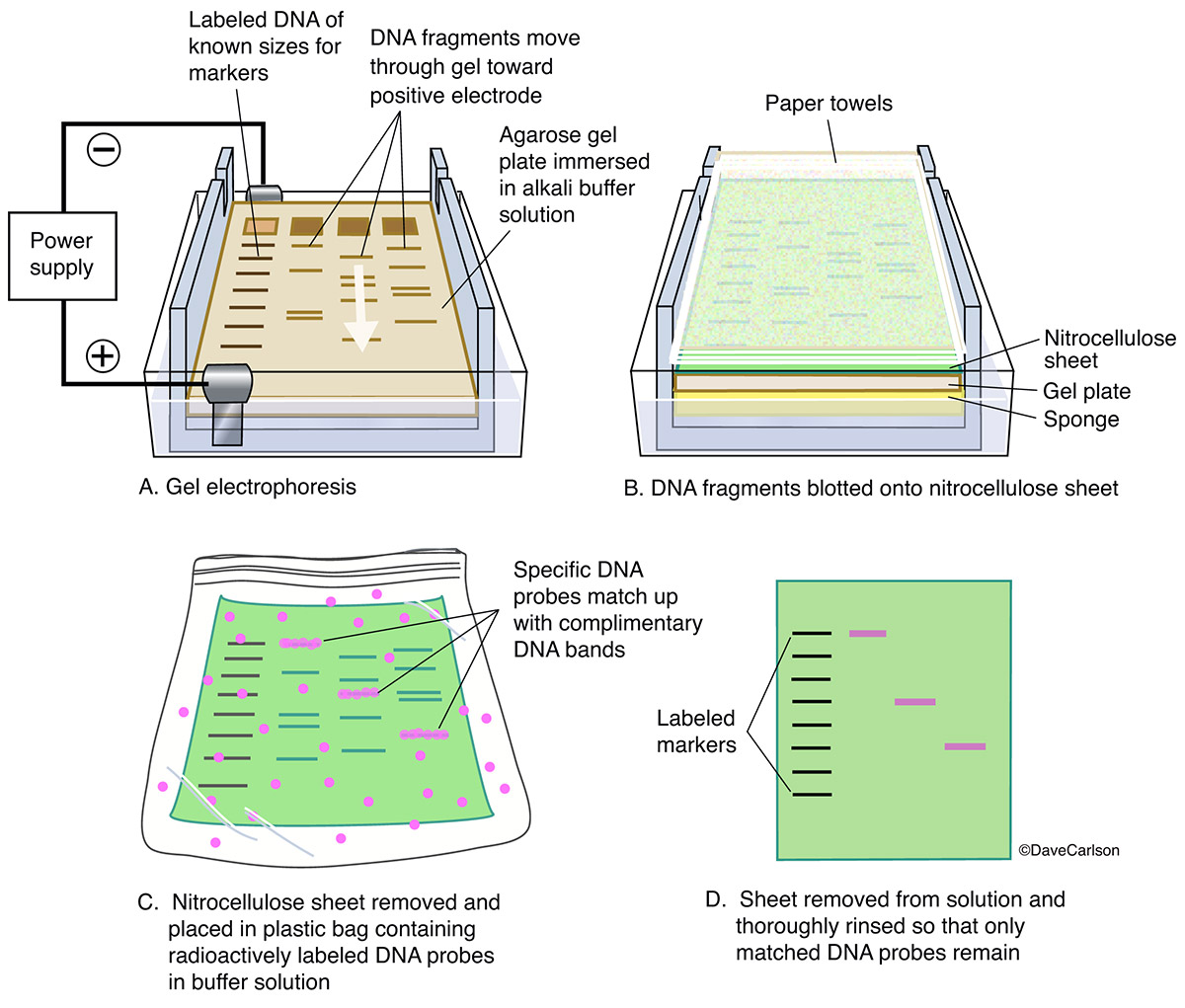
In Western blotting, the molecule of interest is a protein and the probe is typically an antibody raised against that particular protein. It is an Immunoblotting technique that relies on the specificity of binding between a molecule of interest and a probe to allow detection of the molecule of interest in a mixture of many other similar molecules. Western blot: It is applicable for transferring protein andĮastern blot: It is a biochemical technique used to analyze protein post-translational modifications (PTM) and is most often used to detect carbohydrate epitope. Northern blot: It uses for transferring RNA. Southern blot: It uses for transferring DNA. Blot is a technique for transferring DNA, RNA, and proteins onto a carrier so they can be separated, and often follows the use of gel electrophoresis. It is also called immunoblotting and it is a technique for detecting specific proteins separated by electrophoresis by the use of labeled antibodies. No specialized equipment is required for visualization of the colored precipitate, and the produced signal is highly stable.Western blot test is a confirmatory test of HIV-AIDS and it is positive as shown above image. However, colorimetric substrates are perfect for the detection of abundant proteins since the reaction can be monitored visually and allowed to progress until there is adequate color development before being stopped. The limited sensitivity of chromogenic substrates can make it difficult to optimize them for detecting proteins of low abundance, although the chromogenic reaction can be allowed to develop for several hours (or even overnight) to allow the background signal to develop simultaneously. Colorimetric detection relies on the generation of a colored product that becomes deposited on the western blot, which is formed following the conversion of a chromogenic blotting substrate by an appropriate enzyme. The left panel demonstrates indirect detection while the right panel shows direct detection. Schematic representation of colorimetric western blot detection. Each has advantages and disadvantages, which depend on your needs and equipment available in your lab. There are several different choices of readout when western blotting. You can watch our on-demand western blot webinar for more information on the western blot procedure. In direct labeling analysis, the need for the secondary antibody step is eliminated, thereby simplifying the procedure, shortening the protocol, and expediting the time to results.Finally, the membrane is washed again and incubated with an appropriate enzyme substrate (if necessary), producing a reportable signal.The fluorescence of the dye or activity of the enzyme, such as alkaline phosphatase (AP), glucose oxidase (GO) or horseradish peroxidase (HRP), is necessary for signal generation.Following a washing step, the membrane is typically incubated with a dye or enzyme-conjugated secondary antibody that is directed against the primary antibody.After a blocking step, the membrane is probed with a primary antibody that was raised against the antigen in question.In a traditional western blot (indirect labeling), protein samples are first resolved by SDS PAGE and then electrophoretically transferred to the membrane.To learn more about the procedure, refer to our western blot protocol. 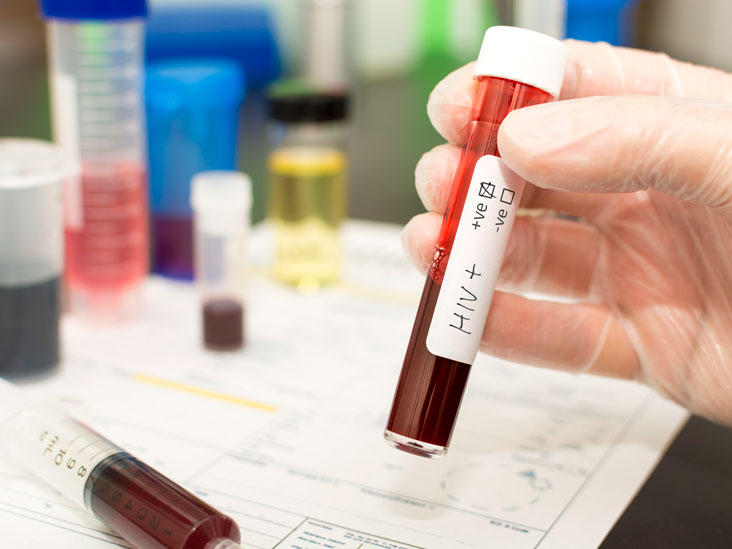
Western blot indirect and direct labelingīefore running a western blot, it is extremely important to research the target protein thoroughly. The western blot technique requires samples to be resolved based on size through sodium dodecyl sulfate-polyacrylamide gel electrophoresis ( SDS PAGE), following which they are transferred to and immobilized on a membrane before antibody-based detection. Western blot aims to identify specific proteins within a complex mixture.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
